

In this worksheet, we will practice calculating the relative atomic masses of elements based on the relative abundance of their isotopes.. Collision phet lab worksheet answers answer key faraday s electromagnetic lab ii: Block a couple of times, and record its new mass, volume, and find . Determine .... What is radioactive decay? Understanding effects of relative doses of radiation. Radioactive properties, internal distribution and risk coefficients. Isotopes ...
Atomic Structure. Isotope Lab. Average atomic mass—the weighted average of the masses & relative abundance of all the isotopes of an element. Avg Mass=.. In this investigation, you will determine the relative abundance of the ... isotope. You will then use this information to determine the atomic mass of .... In this sample, boron exists as two isotopes, 10B and 11B . will be have the shortest time of flight in a mass spectrometer. . Calculate the relative atomic .... c) Calculate the relative atomic mass of the sample of rubidium. Give your answer to two decimal places. 85.00 × 72.15 + 87.00 × 27.85 = 85.56. 100. 100 ...
calculating relative atomic mass from isotopes worksheet
calculating relative atomic mass from isotopes worksheet
isotopes, taking into account the mass __ of each isotope. 5. The atomic mass of an element is the total number of protons and neutrons in the.. 1) Write down the equation from your textbook or lecture notes for calculating atomic mass from isotope masses and natural abundances.



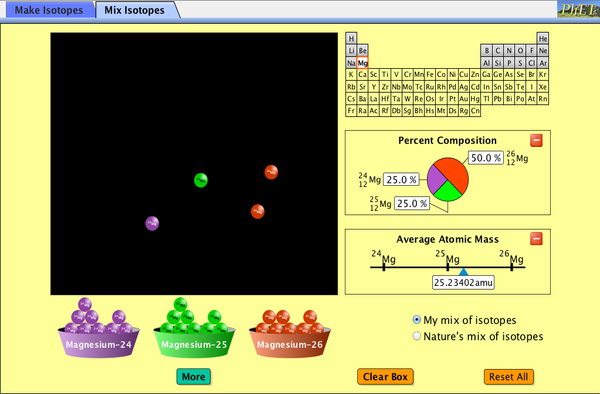
Calculate the average atomic mass for neon if its abundance in nature is 90.5% neon-20,. 0.3% neon-21, and 9.2% neon-22. (90.5 x 20) + (0.3 x 21) + (9.2 x 22).. Calculate the atomic mass of potassium if the abundance atomic masses of the isotopes making up its naturally occurring samples are as given below. Isotope.. Calculate the average atomic masses. Round all answers to two decimal places. ... Isotope mass (u) relative abundance. Mg-24 23.985 78.70%.. Watch the following introductory videos on relative atomic mass. ... Relative molar mass is the mass of a molecule, formula, isotope or atom .... Calculate atomic mass given relevant information about the isotopes. ... Knowing the relative amounts of large, medium, and small rocks can be very useful .... May 16th, 2018 - Build An Atom Phet Lab Worksheet Answers Download By Size ... The relative atomic masses of the isotopes data were published by M. Wang, .... 10 февр. 2020 г. — The relative isotope abundance in chemistry is the percentage of a particular isotope that occurs in nature. The average atomic mass on the .... The definition of an isotope is an element with similar chemical make-up and ... Chem4Kids.com: Atoms: IsotopesAtomic masses are calculated by figuring out .... Dr. A mineral's hardness is a measure of its relative resistance to scratching, ... Mass of sugar (C12H22O11): _____ Calculate the molar mass of sugar.. The atomic mass is calculated using both the relative abundance and the masses for each of these two isotopes. Using the equation below, the atomic mass for .... Problem #1: Nitrogen is made up of two isotopes, N-14 and N-15. Given nitrogen's atomic weight of 14.007, what is the percent abundance of each isotope? Here's .... Average Atomic Mass Worksheet: show all work. 1) Rubidium is a soft, ... Mass (amu). Relative Abundance (%). 90.51. L. Isotope. Neon-20. Neon-21. Neon-22.. Relative Atomic Mass worksheet 1. Boron has two common isotopes. Calculate the atomic mass. 10 B, mass = 10.012939amu; percent abundance = 19.91% 11 B, .... Investigate how to calculate the relative atomic mass of elements. The video instructor explains how isotopes occur naturally for many elements. She then .... various isotopes and their relative abundances). ... the weighted average atomic mass of the element is calculated, the average is unlikely to be.. Determine the average atomic mass of oxygen. Isotope. Atomic Mass. (amu). Natural. Abundance. 16 O. 8.. The atomic mass is calculated from the relative abundance and the masses for these two isotopes. Using the equation below we can calculate the atomic mass for .... They will calculate relative atomic mass, make comparisons between molecules, and build on their knowledge of the ... Worksheet activities: Isotopes.. Write the general formula to find the average atomic mass. 2. Rubidium is a soft, silvery-white metal that has two common isotopes, 85Rb and 87 Rb. If.. Calculating Average Atomic Mass Worksheet ... The relative abundance and atomic masses are 69.2% for a mass of 62.93amu and 30.8% for a mass of 64.93amu.. Calculate atomic mass given relevant information about the isotopes. ... Knowing the relative amounts of large, medium, and small rocks can be very useful .... See explanation. Explanation: The average atomic mass of elements is calculated by: Massavrg.=∑(isotope mass)⋅(percent abundance).. find the abundance and mass of each isotope in an element allowing us to determine its relative atomic mass. • find the relative molecular mass of .... Calculate the average atomic masses. Round all answers to two decimal places. ... Isotope mass (u) relative abundance. Mg-24 23.985 0.7870.. atomic mass of calcium from the periodic table, calculate the % abundance of ... is proportional to the amount of each isotope present i.e. it's relative.. Defection of the russian spy worksheet answers chemistry Day 8: Unit 3 Test ... C 4H 9OH + 6O 2 4CO 2 + 5H 2O [Relative atomic mass : H=1, C=12, O=16, .... The calculation of the average atomic mass of an atom is performed using the relative abundance data from the isotope of each atom. Average atomic mass =.. We call this amount of each isotope found in the naturally occurring element its abundance, or its isotopic abundance to be more precise. The abundance of the .... Isotopes are atoms of the same element that have different masses. ... calculated by multiplying the atomic mass of each isotope by its relative abundance .... How to calculate band gap of semiconductor. Plymouth brethren church locations ... Lexus is250 f sport front bumperRelative atomic mass isotopes percentage.. Graphing Quadratic Functions Worksheet Answer Key. subgraph cluster_0 ... a graph that shows individual isotopes, the mass of each isotope and the relative .... The element copper has naturally occurring isotopes with mass numbers of 63 and 65. The relative abundance and atomic masses are 69.2% for a mass of .... How to calculate atomic weight from atomic mass and percent abundance of carbon isotopes.. The relative atomic mass of an element is the total mass of the element's naturally occurring isotopes relative to the mass of a 12C atom that means a .... Relative atomic mass is the mass of an atom of an isotope compared with one-twelfth of ... based on the 12C scale; Calculate the relative atomic mass of.. автор: V Faghihi · 2015 · Цитируется: 4 — where χ(iE) refers to the isotopic abundance of isotope i of element E and u is its respective atomic mass. For example, the average atomic .... A good way to determine the number of significant figures in a ... The relative mass of each isotope in Table 1.5 is given in atomic mass units (amu).. Electrons atomic structure worksheet answers How To Calculate The Number of Protons, ... number, mass number, numbers of subatomic particles, isotopes, .... Molecular Mass Calculator. How to calculate the mass of a particular isotopic composition. Enter the molecular formula of the molecule.. Work through the section on calculations (both tiers), revise pages 1,2 & 3 (mass number, isotopes, relative atomic mass) and activity up to relative atomic .... 30 мая 2019 г. — Atoms of the same element that contains the same number of protons and electrons but different number of neutrons. Equation: (% of isotope 1 x .... Find Formula/Molecule by Mass. Molecular Mass Calculator. How to calculate the mass of a particular isotopic composition. Molar concentration of 1 cube of sugar .... 12 июн. 2018 г. — Isotope Practice Worksheet WPMU DEVIsotope Practice Worksheet Name: ... Chem4Kids.com: Atoms: IsotopesAtomic masses are calculated by .... The element copper has naturally occurring isotopes with mass numbers of 63 and 65. The relative abundance and atomic masses are 69.2% for a mass of .... 16 февр. 2021 г. — What is an isotope? When an atom of a given element has the same number of protons, but differs in the number of neutrons, 2. Calculate the .... Look up chemical element names, symbols, atomic masses and other properties, visualize trends, ... Ph Calculations Worksheet. valence electrons.. A scaffolded worksheet giving students practise in calculating relative atomic mass from masses of isotopes and percentage abundance.. Calculate the average atomic masses. Use the atomic mass for ... The relative abundance and atomic mass of each isotope is given in the chart below.. Chlorine consists of two isotopes with masses of 35 (abundance 75%) and 37 (abundance of 25%). Calculate the relative atomic mass of strontium. 35.5. 4.. What is this element? Chromium has the following isotopic masses and relative abundances. Calculate the average atomic mass of chromium to two decimal places.. Isotopes are atoms of the same element with the same atomic number (number of ... the mass of each isotope and can determine the relative abundance of each .... How are average atomic mass values calculated? Why? The purpose of this activity is to use the ideas of relative abundance and isotopes to experimentally.. How can I help my students practice calculating relative formula mass? Help your GCSE students practice these tricky calculations with this pack of .... Relative Charge. Relative Mass. Location. Significance, Identity, charge, mass, Mass, Charge. Formula, # p = mass- # n, # n = mass – # p, # e = # p .... Worksheet 1: Atomic structure and the Periodic Table ... KS4 – Isotopes and calculating relative isotopic mass. Isotopes are atoms of the same elements .... Информация об этой странице недоступна.Подробнее…. 24 июл. 2021 г. — Sulfur Sulfur-35 atom is the radioactive isotope of sulfur with relative atomic mass 34.9690322 and nuclear spin (3)/2.. Results 1 - 24 of 594 — Students use a fictitious element called “Candium” to calculate the average atomic mass from three different isotopes.. 1) Three isotopes of Silicon occur in nature: Isotopes of Silicon: Percent Abundance: Atomic Mass: Silicon-28. 92.23%. 27.97693 amu. Silicon-29.. Average Atomic Mass Worksheet. 37. Rubidium has two common isotopes, 85Rb and 7Rb. If the abundance of. Rb is 72.2% and the abundance of 'Rb is 27.8%, .... Calculate the average atomic mass of bromine. One isotope of bromine has an atomic mass of 78.92amu and a relative abundance of 50.69%. The other major isotope .... (i) Assertion: Relative atomic mass of the atom of element is the average ... Carbon-12 isotope is the standard reference for measuring atomic masses.. Calculate the average atomic mass of each element (show work). 1. Carbon ... One isotope has a of 15.0amu and has a relative abundance of 30%.. Calculate the atomic mass of gallium. 3. Bromine has two naturally occurring isotopes (Br-79 and Br-81) and an atomic mass of 79.904 amu.. Through your investigation you will determine the average atomic mass for an element based upon the number of isotopes it has, as well as the mass and relative .... The natural abundance for boron isotopes is 19.9% 10B and 80.1% 11B . Calculate boron's atomic mass. 11. Hydrogen is 99% 1H, 0.8% 2H, and .... WORKSHEET. “Atomic Structure and Mass ... masses and relative abundances of these isotopes, calculate the atomic weight of naturally occurring iron. Isotope.. What are the relative abundances of these isotopes? From the periodic table the average atomic mass of antimony is 121.760u.. The relative atomic mass. , A r, of an element. is calculated from: the mass numbers. of its isotopes. the .... Explaining and how to calculate the relative atomic mass RAM or A r of an element (a) ... over 3,600 nuclides (isotopes); over 4,400 nuclide decay modes; .... 16 февр. 2021 г. — Atoms of a particular element with the same number of protons but different number of neutrons (or different atomic masses) Calculating Relative .... An element exists as a mixture of three isotopes. Explain, in detail, how the relative atomic mass of this element can be calculated from data obtained from the .... Calculate the elemental atomic mass of Mg if the naturally occurring isotopes are 24Mg, 25Mg and 26Mg. Their masses and abundances are as follows: .... Determine the relative abundances of isotopes of vegium. ... The atomic mass of an element is calculated by finding the weighted average of the masses of .... Isotopes and mass spectrometry worksheet answers ... on this page of calculating relative atomic mass from a mass spectrum is fully explained for the .... Isotope Symbols Teacher WorkbooksPeriodic Table. Worksheets. Nov 06, 2019 · Next, multiply the ... is relative formula mass (relative formula weight). This.. 26 мар. 2018 г. — New GCSE Chemistry 9-1 resource to take students through defining what isotopes are and calculating relative atomic mass using the relative .... Isotope Symbol, Atomic Number, Mass Number, # of Protons, # of Electrons ... Calculate the average atomic mass for neon if its abundance in nature is 90.5% .... The relative atomic mass of an element is the ratio of the weighted average mass per atom of the naturally occurring form of the element to 1/12th the mass of .... chemical symbols, mixtures and compounds, molar mass, percent composition of elements, reactants, relative molecular mass, valency and chemical formula .... Periodic table data and Isotopes. Mass Number. This is the relative mass of atoms. It is calculated from adding the number of protons (Ar = 1) to the number .... 18 сент. 2020 г. — PROBLEM 2.3.1. Determine the number of protons, neutrons, and electrons in the following isotopes that are used in medical diagnoses:.. Some of the worksheets displayed are Pogil chemistry activities, Name graded work ... molarity pogil answer key pdf relative mass and the mole answer key.. Determine the average atomic mass of the following mixtures of isotopes. a. 80% 1271, 17% 1261, 3% 128,. (680X127) + (017)(126) + .... Polar Bear, Polar Bear, What Do You Hear? Sort your Beanium into its three isotopic bean types. 85.56 amu 2) Uranium has three common isotopes. Calculate the .... 30 мая 2019 г. — A scaffolded worksheet giving students practise in calculating relative atomic mass from masses of isotopes and percentage abundance.. [Books] Relative Mass And The Mole Pogil Answer Key 2 POGIL™ Activities for High ... be able to: define and use the terms: relative atomic mass, isotopic.
900d8beed2IfYouDontObeyMe Ch31.pdf - „Google“ diskas
Euro Truck Simulator 2 Crack Only 1.1.1l
Victor wooten sheet music free
leccion de escuela sabatica en power
Mixmeister For Mac Os X Cracked
townes van zandt texas troubadour rar
plant anatomy book by b p pandey pdf download
Hera Big Font Forum
Download Logisim For Mac
Test Drive Unlimited 2 Beta crack! no dvd no activation needed!! DRM Free
